 For example, if one photon strikes the electrons, only one electron changes state as a result. Electrons jump between orbitals like particles.The number of electrons orbiting a nucleus can only be an integer.The electron's charge acts like it is smeared out in space in a continuous distribution, proportional at any point to the squared magnitude of the electron's wave function. The electrons are never in a single point location, though the probability of interacting with the electron at a single point can be found from the electron's wave function.Higher energy states are similar to harmonics of that fundamental frequency. 
Thus the lowest possible energy an electron can take is similar to the fundamental frequency of a wave on a string. Electrons do not orbit a nucleus in the manner of a planet orbiting the Sun, but instead exist as standing waves.In this sense, electrons have the following properties: With the development of quantum mechanics and experimental findings (such as the two slit diffraction of electrons), it was found that the electrons orbiting a nucleus could not be fully described as particles, but needed to be explained by wave–particle duality. 8 Electron placement and the periodic table.6.2 Qualitative understanding of shapes.2.3 Modern conceptions and connections to the Heisenberg uncertainty principle.1.1 Formal quantum mechanical definition.The repeating periodicity of blocks of 2, 6, 10, and 14 elements within sections of the periodic table arises naturally from the total number of electrons that occupy a complete set of s, p, d, and f orbitals, respectively, though for higher values of quantum number n, particularly when the atom bears a positive charge, the energies of certain sub-shells become very similar and so the order in which they are said to be populated by electrons (e.g., Cr = 4s 13d 5 and Cr 2+ = 3d 4) can only be rationalized somewhat arbitrarily. In this model the electron cloud of an atom may be seen as being built up (in approximation) in an electron configuration that is a product of simpler hydrogen-like atomic orbitals. Ītomic orbitals are the basic building blocks of the atomic orbital model (or electron cloud or wave mechanics model), a modern framework for visualizing the submicroscopic behavior of electrons in matter. ), omitting j because some languages do not distinguish between the letters "i" and "j". Orbitals for ℓ > 3 continue alphabetically (g, h, i, k. They are derived from the description by early spectroscopists of certain series of alkali metal spectroscopic lines as sharp, principal, diffuse, and fundamental. These names, together with the value of n, are used to describe the electron configurations of atoms. The simple names s orbital, p orbital, d orbital, and f orbital refer to orbitals with angular momentum quantum number ℓ = 0, 1, 2, and 3 respectively. Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. Alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, angular momentum, and an angular momentum vector component ( magnetic quantum number). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. In atomic theory and quantum mechanics, an atomic orbital is a function describing the location and wave-like behavior of an electron in an atom. 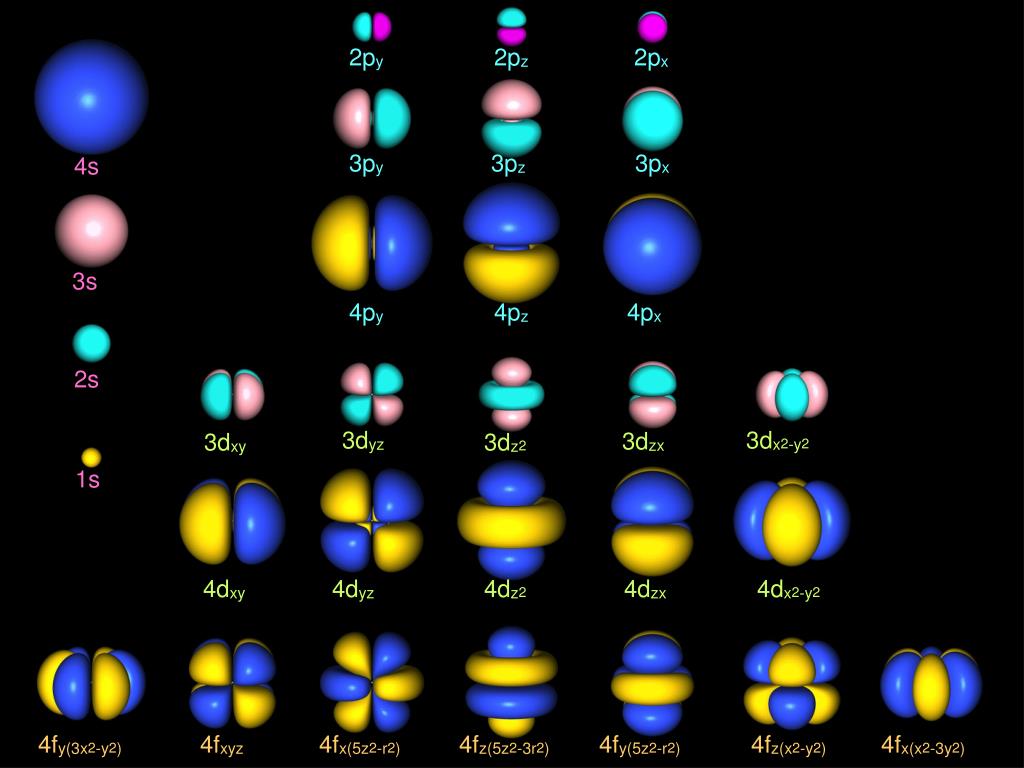
To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depend on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
